July 3, 2025 – Tam Anh Research Institute (TAMRI), in collaboration with Tam Anh General Hospital Group and Sanofi (France), successfully organized an in-depth scientific seminar titled “Breakthroughs in Preventing Invasive Meningococcal Disease.” The event brought together over 3,000 physicians and healthcare experts nationwide to discuss critical advancements in meningococcal disease prevention.

In his opening remarks, Dr. Phuong Le Tri, Executive Director of Tam Anh Research Institute, stated: “We are confronting the continuously evolving epidemiology of meningococcal bacteria – one of the most dangerous infectious agents today.
The advent of the new-generation MenQuadfi vaccine represents a pivotal milestone, extending effective protection to both children and adults, including those over 56 years old – who are accessing this vaccine for the first time in Vietnam. Investing in new-generation vaccine solutions like MenQuadfi is the key to safeguarding and enhancing the health of future generations.”
According to seminar information, the new MenQuadfi meningococcal vaccine, developed by Sanofi in the United States, marks a significant advancement in vaccine technology. The vaccine is notably developed with superior immunogenicity, particularly through its innovative use of tetanus toxoid (TT) as the carrier protein instead of diphtheria toxoid. Global studies confirm MenQuadfi’s effectiveness in stimulating immune responses and reducing the disease burden of meningococcal disease caused by serogroups A, C, Y, and W.
Despite over 40 years of vaccine development, invasive meningococcal disease remains a significant global public health challenge. Approximately 1.2 million new cases are reported annually worldwide, with mortality rates ranging from 10% to 40%.
At the seminar, Dr. Bach Thi Chinh, Medical Director of VNVC, highlighted the severity of invasive meningococcal disease. Caused by Neisseria meningitidis invading the bloodstream or cerebrospinal fluid, it can lead to meningitis, septicemia, and other conditions, progressing rapidly and potentially causing death within hours of symptom onset. Initial symptoms, often flu-like, are hard to recognize but can quickly escalate to severe symptoms such as petechial rash, neck stiffness, and neurological disorders.
According to World Health Organization (WHO) data, Neisseria meningitidis is classified into 12 different serogroups, with A, B, C, X, Y and W being the most prevalent, causing the majority of invasive cases globally. These bacteria commonly colonize the nose and throat of healthy individuals, with carrier rates reaching 5-20%. Adolescents show the highest carriage rates at 23.7%.
Meningitis is the most common invasive form, accounting for about 50% of cases, followed by septic shock (20%) and bloodstream infections (15%). Mortality rates in severe cases can reach 10-15%, while approximately 10-20% of survivors suffer permanent disabilities like hearing loss, seizures, limb amputations and developmental disorders, severely impacting their quality of life and that of their families.


Dr. Chinh emphasized that meningococcal bacteria pose a global public health challenge due to their high rates of complications, critical illness, mortality, and permanent sequelae – even with early detection and timely treatment.
Analysis of 35 meningococcal outbreaks across six global regions from 1973 to 2018 reveals continuous shifts in dominant serogroups over time. From 1970 to 1980, outbreaks were predominantly caused by serogroups A and B, shifting to A and C in the early 2000s, then to B and C in the latter half of that decade. Notably, since 2016, serogroup W has emerged as a significant epidemiological factor.
In Turkey, from 2005 to 2008, serogroup B increased from 40% to 57% of cases, while by 2012 serogroup W became predominant at 67%. Meanwhile, in 2023, serogroup Y showed a sudden surge in the U.S., emerging as a newly significant serogroup in recent decades.
“Interestingly, the intervals between outbreaks are becoming shorter; previously outbreaks were separated by months or even years, whereas now this interval can be less than six months,” noted Dr. Chinh.
In Vietnam, recent studies show serogroup B predominates among hospital-reported cases, while serogroup C (7%) and W are also present.
Dr. Chinh emphasized: “The continuous changes in circulating meningococcal serogroups present major challenges for accurate epidemiological assessment. Combined with evolving diagnostic and treatment methods, this has become a significant barrier to disease surveillance and management. Consequently, developing prevention and control strategies is increasingly complex and requires flexible adaptation.”
Furthermore, meningococcal serogroups do not provide cross-immunity – vaccination against one serogroup (e.g., A) doesn’t protect against others (B, C, W, Y) – necessitating active immunity development against all circulating serogroups for optimal prevention.
Therefore, Dr. Chinh noted that multivalent vaccines are expected to enhance coverage of circulating meningococcal serogroups, exemplified by the quadrivalent MenACYW vaccine.
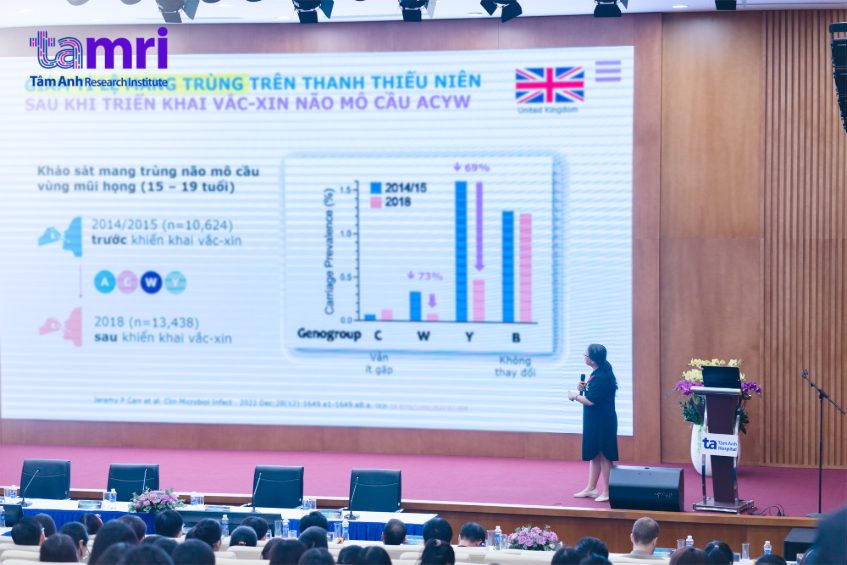
The new conjugate meningococcal vaccine, utilizing tetanus toxoid as a carrier protein, demonstrates strong immunogenicity and safety, and contributes to reducing asymptomatic carriage within the community.
Since 2011, health experts have worked to reduce meningococcal disease impact through improved rapid diagnosis, community surveillance, and public awareness. Vaccination, particularly with quadrivalent MenACYW vaccines, plays a paramount role in prevention and control. Vaccination efforts now extend beyond high-risk groups (infants, children, and adolescents who have high carriage, transmission and mortality rates) to include adults, elderly, and those with underlying conditions, strengthening community protection and preventing disease spread.
The field is witnessing a critical vaccine technology advancement with the replacement of diphtheria toxoid carrier protein (CRM197) with tetanus toxoid (TT). “The quadrivalent MenACYW-TT vaccine, also called MenQuadfi, uses advanced technology combining polysaccharides with tetanus toxoid carrier protein, offering superior benefits including enhanced immune response, improved immune cell activity, and strengthened long-term immune memory,” Dr. Chinh added.
Dr. Nguyen An Nghia, Deputy Head of Infectious-Neurology Department at Children’s Hospital 1 in Ho Chi Minh City, explained that MenQuadfi’s conjugate technology provides not only superior immunogenicity but also enhances immune memory – particularly beneficial for children under 2 years who respond poorly to plain polysaccharide vaccines.
“As of April 2025, with over 20 million doses administered and licensed in more than 70 countries, MenQuadfi has demonstrated both safety and high immunogenicity,” Dr. An Nghia noted.
A Finnish study evaluated MenQuadfi’s immunogenicity and safety, showing protective antibody levels in 12-23 month-olds, with particularly outstanding efficacy against serogroup C and robust immune response post-vaccination, indicating strong protective capacity.
Similarly, in adolescents, MenQuadfi showed high protective efficacy and immunogenicity when co-administered with other vaccines like Gardasil 9 (HPV vaccine) and Tdap-IPV (diphtheria-tetanus-pertussis-polio vaccine) without compromising immune response.
Notably, in adults ≥56 years, MenQuadfi induces better immune response than plain polysaccharide vaccines, creating vaccination opportunities for this previously unvaccinated high-risk group (who face highest complication and mortality rates). Moreover, it can be co-administered with MenB vaccines without efficacy reduction, optimizing vaccination schedules and strengthening community protection.
Through controlled clinical studies, Dr. An Nghia affirmed: “Clearly, MenQuadfi is not only safe but provides superior immune response, particularly against serogroup C. This promises major advances in vaccinating children, adolescents, and the elderly.”


Dr. An Nghia stated that the MenQuadfi vaccine has demonstrated a good safety profile and strong immunogenicity, even when co-administered with other vaccines such as the 9-valent HPV vaccine or Tdap-IPV.
MenQuadfi plays a crucial role in generating T-cell-dependent immune response – essential for immune memory formation and booster effectiveness. This advantage surpasses plain polysaccharide vaccines which don’t produce immune memory.
Additionally, MenQuadfi’s molecular structure features optimized linkers that increase surface area, facilitating antigen recognition and more effective immune response. The vaccine’s stable antigen-carrier protein linkage with cross-linked structure ensures durability while enhancing optimal immune stimulation.
In summary, the new-generation MenACYW vaccine (MenQuadfi) provides not only superior protection but also high safety and stability. Designed for flexible administration, it can be co-administered with other critical vaccines, optimizing immunization schedules against dangerous infectious diseases.

At the conclusion of the program, experts emphasized that the next generation ACYW-TT meningococcal vaccine, MenQuadfi, has shown superior immune response and preventive efficacy.
LATEST UPDATE: July 4, 2025 VNVC Vaccination Center System has officially introduced and begun administering the new-generation MenACYW-TT meningococcal vaccine (MenQuadfi) for children and adults at hundreds of VNVC centers nationwide. This marks Vietnam’s first-ever meningococcal vaccination access for adults aged 56+, protecting this high-risk group against the deadly disease that most severely affects young children, adolescents, and the elderly.
Tam Anh Research Institute
Tam Anh Research Institute